Question 1:
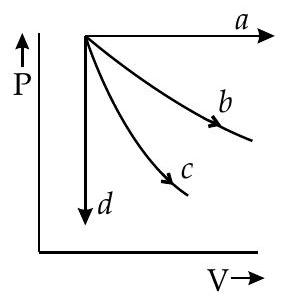
The given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
A)
B)
C)
D)
Answer:
Solution:
In an isochoric process, volume remains constant while in isobaric process, pressure remains the same.
Slope of an isothermal process is given as
while for an adiabatic process, slope of curve is , where .
Adiabatic curve is steeper than isothermal curve.
Prev Exam(s):
JEE MAIN
- 2019
, 8 Apr Shift-02
Chapter:
THERMODYNAMICS
Question Level:
L2
Tags:
- THERMODYNAMICS
- PHYSICS



