Question 1:
1 kg of water at 100 °C is converted into steam at 100 °C by boiling at atmospheric pressure. The volume of water changes from 1.00×10-3 m3 as a liquid to 1.671 m3 as steam. The change in internal energy of the system during the process will be (Given latent heat of vaporization = 2257 kJ/kg, Atmospheric pressure = 1 × 105 Pa)
A)
B)
C)
D)
Answer:
Solution:
Given m=1 kg,
Change in volume at constant pressure and temp:
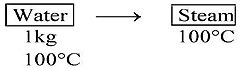
Using first law of thermodynamics
Using
Prev Exam(s):
JEE MAIN
- 2023
, 11 Apr Shift-01
Chapter:
CALORIMETRY
Question Level:
L2
Tags:
- PHYSICS
- CALORIMETRY



